Introducing Howard Chen, physician and fire safety director for Flow Arts Institute. Together, we’ve created a Fire and Fuel safety program. The content is split into 3 parts: Fire Performer Safety for those dancing with fire, Fire Safety Personnel for those keeping the dancers safe, and Fuel Safety for safe protocols for managing your fuel spin off, storage and disposal. There is a manual and future in-person trainings available.
The first content video of the series introduces Fire Science. It deals with the science of the element of fire which is vital to Fire Performers and Safety Personnel understanding.
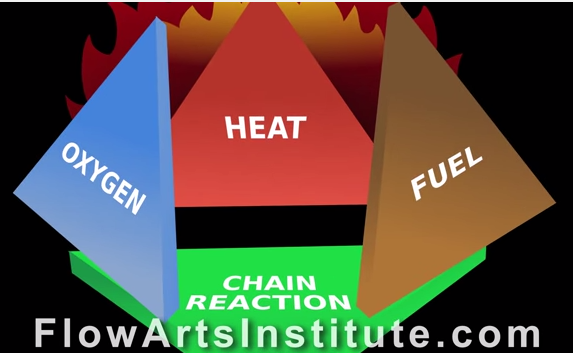
What really is fire? Fire is scientifically defined as the rapid oxidation of material in the exothermic chemical process of combustion, releasing heat and light among other reaction products. But in common terms this means that fire is a chemical reaction that involves oxygen that gives of things like heat, light and other byproducts including soot and smoke. Actually, heat and oxygen are essential components to fire that make up 2 sides of the fire triangle. The last side, of course, is a combustible fuel. The fire triangle is simplistic, but an accurate representation of the components necessary to start a fire. Once a fire is lit, a better graphical representation of an ongoing fire is the fire tetrahedron which includes the three components of the triangle, but also indicates that the chemical chain reaction of the three components is essential to the continuation of the chemical reaction that we know as fire.
It is important to know the difference between fire and flame. Flame is the visible manifestation of fire with the color of the flame being determined by multiple factors, including the fuel that is used, the amount of oxygen, and the heat of the fire. For instance, a yellow flame indicates an incomplete combustion where there is an imperfect ration of oxygen, heat, and fuel. While a blue flame indicates complete combustion where the oxygen, heat, and fuel are all in a perfect ratio to complete the chemical reaction. Flame color also gives us an idea of the temperature of the flame that we are dealing with. For instance, during a fire performance, the hotter white, yellow and orange flames occurring at the beginning of the burn with cooler red flames produced when the fuel is being exhausted. Color flames can also be made by burning different fuel combinations. For instance, a common, albeit unhealthy combination, includes the use of a denatured alcohol fuel mixed with boric acid which produces a green flame.
Now for a review, fire is a chemical reaction which requires a combination of oxygen, heat, and fuel. Once a fire is ignited, chain reactions between the three components continue the chemical reaction we know as fire. Finally, flame colors can indicate the completion of the combustion process as well as the temperature of the flame. And finally, as a reminder, no matter how much we understand about fire, it is still quite a dangerous and destructive process. For instance, during a fire performance, at midburn, the wicks come close to a temperature of 1000 degrees Fahrenheit, certainly enough to cause a significant burn on exposed flesh. For more information on how to keep safe, tune into our next video. We will be featuring these videos the last Monday of every month.